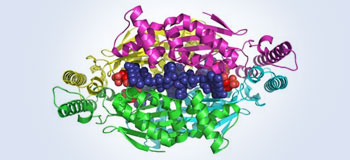
 Chemical reaction for DNA production in bacteria and virus
A team of researchers has discovered a new chemical reaction for producing one of the four nucleotides, or building blocks, needed to build DNA.
The reaction includes an unusual first step or mechanism (unlike other known reactions that produce the DNA building block). It uses an enzyme (flavin–dependent thymidylate synthase, or FDTS) that speeds up, or catalyzes, the reaction without bonding to any of the compounds,
or substrates, in the reaction. The enzyme is coded by the thyX gene and has been found primarily in bacteria and viruses,
including several human pathogens and biological warfare agents.
In the future, scientists may use this knowledge for the development of new anti–bacterial and anti–viral drugs.
Drug development
Researchers has devised a new method for making complex molecules.
The reaction they have come up with should enable chemists to synthesize new varieties of a whole subclass of organic compounds called
nitrogen–containing heterocycles, thus opening up new avenues for the development of novel pharmaceuticals
and natural products ranging from chemotherapeutic compounds to bioactive plant materials such as morphine.
Thermite reaction process in railway welding (shortly after this, the liquid iron flows into the mould around the rail gap).
Chemical reactions are central to chemical engineering where they are used for the synthesis of new compounds from natural raw materials such
as petroleum and mineral ores. It is essential to make the reaction as efficient as possible, maximizing the yield and minimizing the amount of reagents,
energy inputs and waste. Catalysts are especially helpful for reducing the energy required for the reaction and increasing its reaction rate.
Chemical reaction for DNA production in bacteria and virus
A team of researchers has discovered a new chemical reaction for producing one of the four nucleotides, or building blocks, needed to build DNA.
The reaction includes an unusual first step or mechanism (unlike other known reactions that produce the DNA building block). It uses an enzyme (flavin–dependent thymidylate synthase, or FDTS) that speeds up, or catalyzes, the reaction without bonding to any of the compounds,
or substrates, in the reaction. The enzyme is coded by the thyX gene and has been found primarily in bacteria and viruses,
including several human pathogens and biological warfare agents.
In the future, scientists may use this knowledge for the development of new anti–bacterial and anti–viral drugs.
Drug development
Researchers has devised a new method for making complex molecules.
The reaction they have come up with should enable chemists to synthesize new varieties of a whole subclass of organic compounds called
nitrogen–containing heterocycles, thus opening up new avenues for the development of novel pharmaceuticals
and natural products ranging from chemotherapeutic compounds to bioactive plant materials such as morphine.
Thermite reaction process in railway welding (shortly after this, the liquid iron flows into the mould around the rail gap).
Chemical reactions are central to chemical engineering where they are used for the synthesis of new compounds from natural raw materials such
as petroleum and mineral ores. It is essential to make the reaction as efficient as possible, maximizing the yield and minimizing the amount of reagents,
energy inputs and waste. Catalysts are especially helpful for reducing the energy required for the reaction and increasing its reaction rate.
A chemical reaction is a process that leads to the transformation of one set of chemical substances to another. The substances, initially involved in a chemical reaction are called reactants or reagents. Reactions often consist of a sequence of individual sub–steps are the so–called elementary reactions. The information on the precise course of action is part of the reaction mechanism.
Synthesis, Decomposition, Single replacement and Double replacement are four different types of elementary reactions. Redox reactions can be understood in terms of transfer of electrons from one involved species (reducing agent) to another (oxidizing agent). Acid–base reactions involve transfer of protons from one molecule (acid) to another (base). Here, acids act as proton donors and bases as acceptors. The associated proton transfer results in the so–called conjugate acid and conjugate base. The reverse reaction is possible, and thus the acid/base and conjugate base/acid are always in equilibrium.
The speed or rate of chemical reactions is controlled by five factors: the nature of reactants, the ability of reactants to meet, concentration of the reactants, temperature and the presence of catalysts. According to collision theory, the rate of a reaction depends on the number of effective collisions per second of the reactant particles which is only a small fraction of total number of collisions per second. The molecular kinetic energy called activation energy and temperature affect the rate of reaction. The detailed sequence of elementary processes that lead to the net chemical change is the mechanism of reaction.
Catalysts change reaction rate but are not consumed by the reaction.
Chemical thermodynamics is the study of the role of energy in chemical changes and in determining the behavior of materials. Enthalpy is a measure of the total energy of a thermodynamic system. It includes the internal energy, which is the energy required to create a system, and the amount of energy required to make room for it by displacing its environment and establishing its volume and pressure.
The unit of measurement for enthalpy in the International System of Units (SI) is the joule, but other conventional units are still in use, such as the British thermal unit and the calorie.
The first law of thermodynamics states that internal energy may be transferred as heat or work but it cannot be created or destroyed. A spontaneous change is a change that continues without outside intervention and spontaneous processes tend to proceed from states of lower probability to states of higher probability. The higher probability states are those that allow more options for distributing energy among the molecules. So we can also say that spontaneous process tends to disperse energy.
Entropy is a measure of the number of equivalent ways to distribute energy in the system. If energy were money, entropy would describe the number of ways of counting it. An increase in freedom of molecular motions corresponds to an increase in entropy. All spontaneous processes increase the total entropy of the universe. The second law of thermodynamics states that all real processes increase the total entropy of the universe. The third law of thermodynamics states that at absolute zero temperature the entropy of a perfectly ordered pure crystalline substance is zero. The third law of thermodynamics makes experimental measurement of absolute entropies possible.
As we know the point at which entropy has value of zero, it is possible to measure the total amount of that a substance has at temperature above zero degree Kelvin (0 K). The entropy of one mole of substance at a temperature of 298 K and a pressure of 1 atm is called the standard entropy which is used to calculate standard entropy of change for chemical reactions.
The field of electrochemistry is one of the many areas in which the principles of thermodynamics lead to practical benefits. An electrochemical cell uses electrical energy to drive a non–spontaneous chemical reaction. Oxidation and reduction occur at anode and cathode respectively to facilitate conduction. Batteries are used in a variety of applications including a mobile phone or a pace maker used by a heart patient to maintain a reasonable heart rate. Batteries are designed by applying principles of electrochemistry.