When both the reactants are same : This means that two molecules of the same reactant are involved in the chemical reaction. The second‐order reaction in this case would be represented as
2A → P
and the rate of the reaction would be expressed as,

where ‘a’ is the initial concentration of ‘A’, ‘x’ is the concentration of the product formed after time ‘t’ and ‘(a − x)’ is the concentration of ‘A’ remaining at time ‘t’.
Separating the variables and integrating, we have
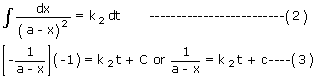
Knowing that at t = 0, x = 0 we have C =
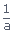 .
.
Hence,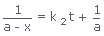
Transposing and solving for k2, we get
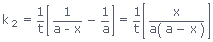 ‐ ‐ ‐
‐
‐ ‐ ‐ ‐ ‐
‐ (4)
‐ ‐ ‐
‐
‐ ‐ ‐ ‐ ‐
‐ (4)
It is the required integrated expression for the rate constant of a second‐order reaction in which two molecules of the same reactant are involved in the reaction.
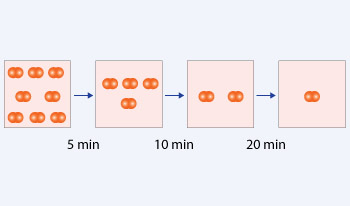
In the above reaction, initial concentration is 8 molecules. Time taken to consume 4 molecules is 5 minutes which is the half–life of the reaction. After first half–life, 10 minutes is taken to consume half the molecules i.e. 2. After second half–life, 20 minutes is taken to consume half the molecules i.e. 1. The half–life is inversely proportional to the initial concentration. Hence, it is a second order reaction.