Oxoacids of phosphorous
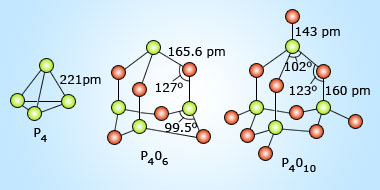
Phosphorous(III)oxide, P4O6,is obtained by allowing white phosphorous to oxidize in a limited supply of oxygen. When oxidation takes place in the presence of excess oxygen, phosphorous(V)oxide, P4O10, forms. This compound is also readily formed by oxidation of P4O6. These two oxides represent the two most common oxidation states for phosphorous, +3 and +5.
Phosphorous (V) oxide is the anhydride of phosphoric acid, H3PO4, a weak triprotic acid. In fact, P4O10 has a very high affinity for water and is consequently used as a drying agent. Phosphorous(III) oxide is the anhydride of phosphorous acid, H3PO3,a weak diprotic acid.