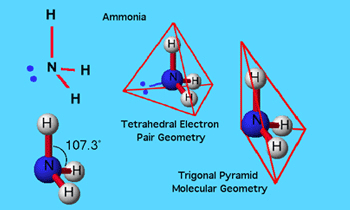
 Structure Of Ammonia
Showing the tetrahedral arrangement, with the three 'H' atoms and lone
pair of electrons occupying the four tetrahedron corners.
Structure Of Ammonia
Showing the tetrahedral arrangement, with the three 'H' atoms and lone
pair of electrons occupying the four tetrahedron corners.
First, we need to remember that the nitrogen, in addition to forming three sigma bonds, also carries an unshared electron pair.This means that there are four groups of electrons associated with the nitrogen.Mutual repulsion of these groups leads to a tetrahedral arrangement, much like that of a typical sp3 carbon atom.This predicts a bond angle between the N-H bonds of 109.5° which agrees pretty well with the observed value of 107° found in ammonia.
First, we need to remember that the nitrogen, in addition to forming three sigma bonds, also carries an unshared electron pair.This means that there are four groups of electrons associated with the nitrogen.Mutual repulsion of these groups leads to a tetrahedral arrangement, much like that of a typical sp3 carbon atom.This predicts a bond angle between the N-H bonds of 109.5° which agrees pretty well with the observed value of 107° found in ammonia.