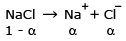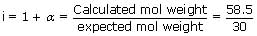In certain solvent, phenol dimerises to the extent of 60%. Its observed molecular mass in the solvent should be
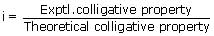
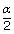 ∴ 0.7
∴ 0.7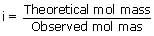
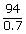
A solution containing 8.6 g urea in one litre was found to be isotonic with a 5% (wt./vol.) solution of an organic non – volatile solute. The molecular weight of latter is
For isotonic solution
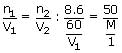
⇒ M = 348.9
Osmotic pressure of blood is 7.55 atm at 310 K. An aqueous solution of glucose that will be isotonic with blood is...........wt/vol
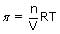
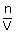 times; 0.0821 × 310
times; 0.0821 × 310
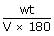 × 0.0821 × 310
× 0.0821 × 310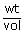 = 54.1 g/lt = 5.41%M
= 54.1 g/lt = 5.41%M The relationship between the values of osmotic pressures of 0.1 M solutions of KN03(P1) and CH3COOH (P2) is
The mol. weight of NaCl determined by studying freezing point depression of its 0.5% aqueous solution is 30. The apparent degree of dissociation of NaCl is