Isothermal and Adiabatic changes
| S.No | Isothermal Change | Adiabatic Change |
|---|---|---|
| 1. | Temperature remains constant, i.e Δ = 0 | No exchange of heat ΔQ = 0 |
| 2. | System is thermally conducting to the surroundings | System is thermally insulated from the surroundings |
| 3. | The process takes place slowly | The process takes place quickly |
| 4. | Internal energy remains constant ΔU = 0 | Internal energy changes ΔU ≠ 0 |
| 5. | Specific heat capacity is infinite | Specific heat capacity is zero |
| 6. | Equation of state is PV = constant | Equation of state is PVγ = constant |
| 7. |  |
 |
| 8. | Isothermal elasticity Eθ = P | Adiabatic elasticity Eϖ = γP |
| 9. |  |
 |
Comparison between isothermal and adiabatic curves:
When expanded to the same volume from the same initial state.
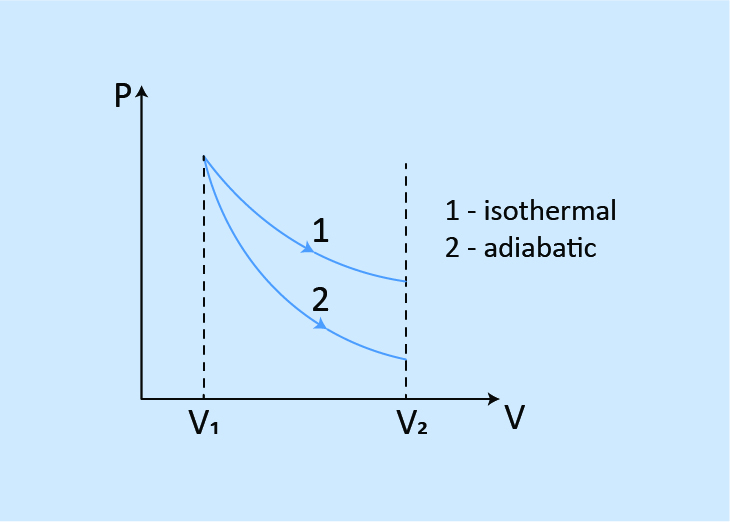
Work done: W1 > W2
Final pressure: P1 > P2
Final temperature: T1 > T2
When compressed to the same volume from the same initial state
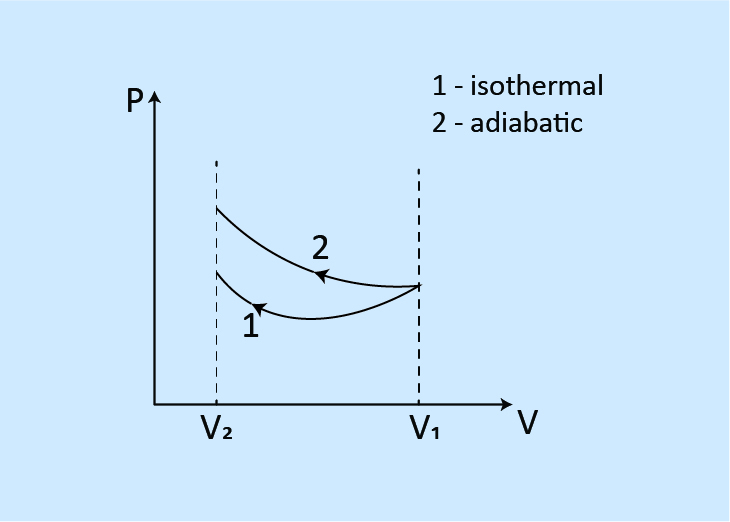
Work done: W1 < W2
Final pressure: P1 > P2
Final temperature: T1 > T2
When expanded to the same pressure from the same initial state.
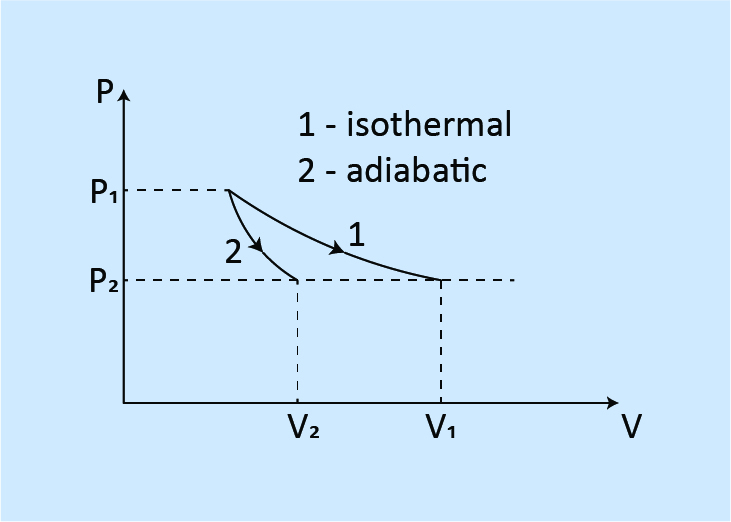
Work done: W1 > W2
Final volume: V1 > V2
Final temperature: T1 > T2
When compressed to the same pressure from the same initial state.
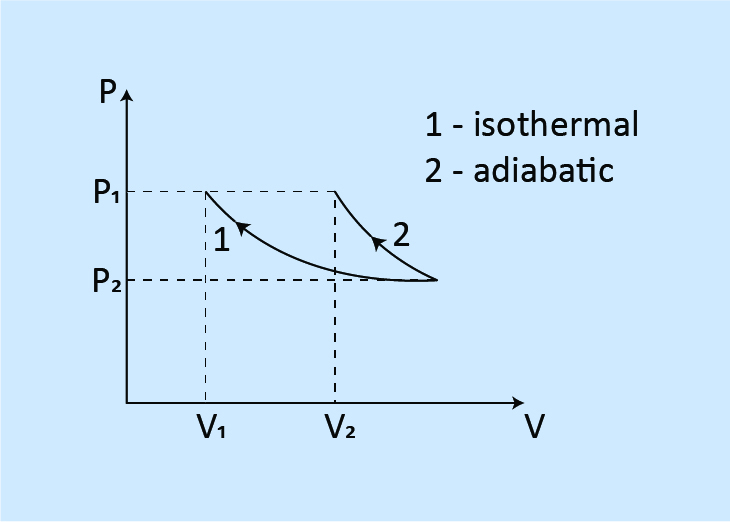
Work done: W1 < W2
Final volume: V1 < V2
Final temperature: T1 < T2