Relation between equilibrium constant and energy
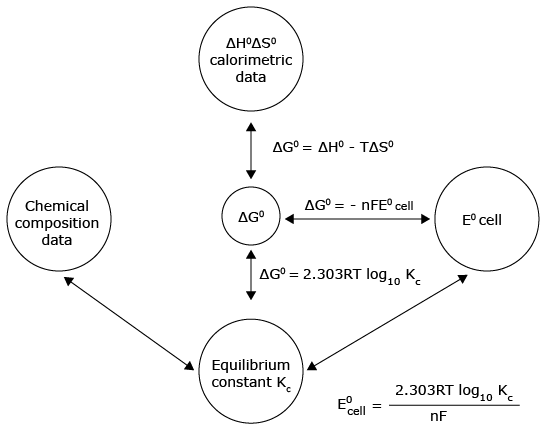
The standard free energy change, ΔG° = –2.303RTlog10K, where K is
equilibrium constant.
(a) Thus if K > 1, then ΔG° = –ve thus reactions with equilibrium constant k
> 1 are thermodynamically spontaneous
(b) If K < 1, then ΔG°=+ve and thus reactions with equilibrium constant K < 1 are thermodynamically spontaneous in reverse direction.
The electrical work is equal to the product of the EMF of the cell and electrical charge that flows
through the external circuit i.e.,
Wmax = nFecell.....(i)
According to thermodynamics the free energy change (ΔG) is equal to the maximum work. In the cell work is done on the surroundings by which electrical energy flows through the external circuit, So
–Wmax = ΔG .....(ii)
from eq.(i) and (ii) ΔG = –nKEcell
In standard conditions ΔG° = –nFE°cell
Where ΔG° = standard free energy change
But 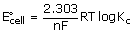
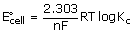
∴ 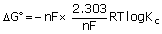
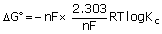
ΔG° = –2.303 RT log Kc or ΔG = ΔG° + 2.303 RT log Q
ΔG° = –RT ln Kc (2.303 log X = ln X)
Relation amongst equilibrium constant and partial pressure
Consider a general cell reaction involving n electrons.
aA + bB ⇌ cC + dD
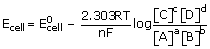
Concentration of solids and liquids is taken as unity, concentration of ions Mol L–1
and concentration of gasses as partial pressures in atmosphere.
Relation amongst equilibrium constant and mole fraction
When Ecell drops to zero the concentration of ions will be equilibrium concentrations. We
have for general cell reaction
aA + bB ⇌ cC + dD
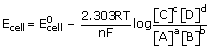
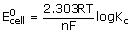 [since at
equilibrium Ecell = 0]
[since at
equilibrium Ecell = 0]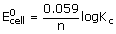 at 298 K.
at 298 K.Q
The ΔG0 for the reaction
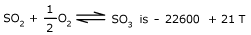
Determine the temperature at which mixture is 80 % (by mole) in SO3, if initial
mixture contain 15 % SO2 and 20 % O2 at one atmosphere total
pressure. Assume total pressure is maintained constant at one atmosphere throughout.
Sol:
Let initially, there are 100 moles of gaseous mixture. Then
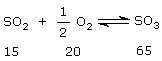
After changing temperature to T, let x mole of SO2 is consumed.
Now at new equilibrium : SO2 = 15 – x
O2 = 20 – (x/2)
SO3 = 65 + x
From the given condition : 65 + x = 0.8 (100 – (x/2))⇒ x =
10.7
Therefore,partial pressure at new quilibrium are :
SO3 = (75.7/94.65) atm, SO2 = (4.3/94.65 ), O2 = (14.65/94.65)
Kp (T) = (75.7/94.65 ) × (94.65/4.3) × ((94.65/14.65)) = 44.75
Also,ΔG0 = – RT ln Kp = – 22600 + 21 T ⇒ ln
Kp = (22600/RT ) – (21/R ) = 3.8
Solving, T = 429.7K