Fac-mer isomers of geometric isomerism
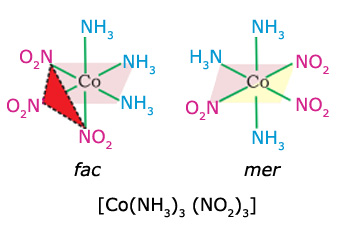
In octahedral complex [of the type Ma3b3] three donor atoms of the same ligands occupy adjacent positions at the corners of an octahedral face, to form facial (fac) isomer. When the positions are around the meridian of octahedron, it forms meridional (mer) isomer. For example, fac-mer isomers of [Co(NH3)3(NO2)3] complex.
Note:
Tetrahedral complexes do not exhibit geometrical isomerism as well as fac-mer because the relative positions of the unidentate ligands on the central metal atom are the same with respect to each other.