Enthalpy of hydration of copper sulphate
Enthalpy of hydration of anhydrous CuSO4 cannot be determined
directly.
CuSO4(s) + 5H2O(l) →
CuSO4.5H2O(s) ΔH3
But it is possible to measure the change in enthalpy when anhydrous CuSO4
and hydrated
CuSO4 are dissolved in a large excess of water separately.
Using these values, and applying Hess law, it is possible to calculate the enthalpy
change in
the required reaction above
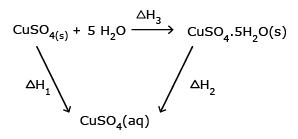
Applying Hess' law:
ΔH1 = ΔH3 + ΔH2
ΔH1 – ΔH2 = ΔH3
Procedure:
Note:The masses of compounds chosen are chosen such that they have
approximately the
same number of moles and give approximately the same molarity solution when dissolved.
Part I
- Accurately weigh out approximately 5g of anhydrous copper(II) sulfate (Mol wt = 159.5) using a weighing bottle
- Measure out 50 ml water into a weighed polystyrene beaker, re-weigh the beaker + water and record the temperature of the water over a couple of minutes
- Quickly add the anhydrous copper(II) sulfate to the water and stir until dissolved. Record the highest temperature reached and, hence, the temperature change, ΔT.
- Repeat the test several times.
Part II
- Accurately weigh out approximately 7.8 g of copper(II) sulfate pentahydrate (M.wt = 249.5)
- Measure out 50 ml water into a weighed polystyrene beaker, re-weigh the beaker + water and record the temperature of the water over a couple of minutes
- Quickly add the copper(II) sulfate pentahydrate to the water and stir until dissolved. Record the final temperature reached and, hence, the temperature change, ΔT
- Repeat the test several times
Observations:
The energy change for each trial can be calculated from the formula:
Enthalpy change = mass of water (kg) × specific heat capacity of water ×
temperature
change.
ΔH = m × c × ΔT
Energy per mole = energy change/ number of moles of salt used
Energy per mole = ΔH/moles
This can then be used to find out the enthalpy change of the reaction:
CuSO4(s) + 5H2O(l) →
CuSO4.5H2O(s)
Applying Hess' law:
ΔH(anhydrous salt) – ΔH(hydrated salt) = ΔH(reaction)