Crystal Field Splitting
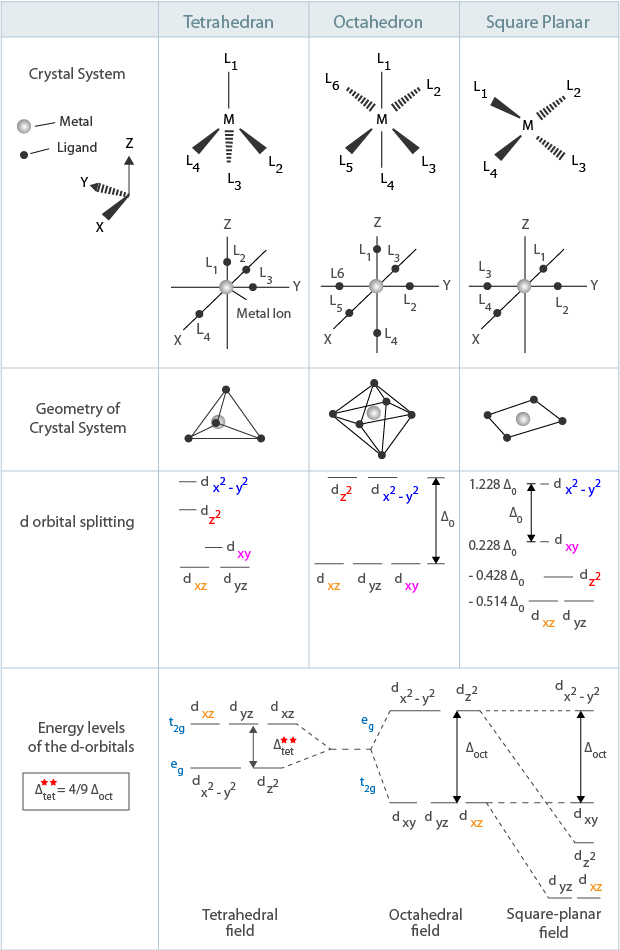
Limitation associated with the crystal field splitting is that it does not consider the affect of covalent bonding between the ligand and the metal atom. It focuses on negative charged ligands as point charges that occupy the last position in series. This may deviate from the reality of the properties exhibited by metal in a complex that are explained by ligand field and molecular orbital theories.