 Fuel cells could be the next generation energy–saving technology to generate electrical power for our homes and cars. The biggest advantage of fuel cell is that it makes power more efficiently and with less pollution. A fuel cell typically converts the chemical energy of its fuel into electricity with an efficiency of about 50%.
Fuel cells could be the next generation energy–saving technology to generate electrical power for our homes and cars. The biggest advantage of fuel cell is that it makes power more efficiently and with less pollution. A fuel cell typically converts the chemical energy of its fuel into electricity with an efficiency of about 50%.
Fuel cells are ideally emission–free as compared to common fuels such as methane or natural gas that generate carbon dioxide. The problem is that coal is a relatively dirty energy source, which creates lot of pollution and wastage. Fuel cells on the other hand convert the chemicals hydrogen and oxygen into water, and in turn produce electricity with very little pollution.
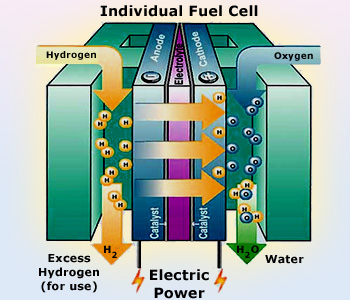
A fuel cell is an electromechanical device that coverts chemical energy into electricity. It combines hydrogen fuel with oxygen producing electricity, heat and water. The reaction occurs as long as fuel is available. In this manner, a fuel cell is similar to a battery.
The oxygen is taken from air while hydrogen is stored in pressurized containers. There are no harmful emissions at all due to the absence of combustion - the process of converting carbon fuel to energy. And the by-product is pure water. (So clean is the water emitted that at one of the power system company, visitors were served hot tea made with it!).
A fuel cell can be considered to be electrolysis in reverse. Like in electrolysis, fuel cell has also two electrodes separated by an electrolyte. The negative electrode (or anode) receives hydrogen while the positive electrode (or cathode) collects the oxygen. Hydrogen is separated into positively charged H+ ions and electrons by the catalyst at the anode. The oxygen is ionized and migrates to the anode compartment through the electrolyte. Here it combines with hydrogen.
Typically, a single fuel cell produces 0.6 - 0.8V under load conditions. Several such fuel cells can be stacked i.e connected in series to obtain higher voltages of the order of several hundred kilowatts(kW).
(Refer to animation)
Hydrogen (the simplest chemical element with one proton and one electron) makes up 90% of the universe. It is the third most abundant element on earth's surface (after nitrogen and oxygen). So with very easily available raw material, the cost of conversion into electrical energy should be relatively low. But it is not so due to hydrogen being strongly bonded to other substances. It takes considerable energy to separate and get hydrogen gas. In terms of net calorific value (NCV), it is more costly to produce hydrogen gas than gasoline.