 Various elements
Various elements
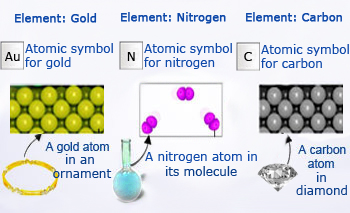
An element consists of only one kind of atoms. For example, pure gold. when a bit of golden ornament (made of pure gold) is analyzed by a powerful microscope, it was observed that a chain of gold atoms arranged in systematic manner to have the shiny appearance. Similarly when a flask of gaseous nitrogen is observed, it has only nitrogen atoms. In case of diamond, carbon atoms are arranged to have gaint structure.
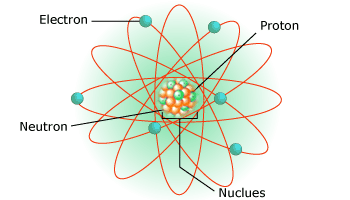
Protons, neutrons and electrons are the subatomic particles that are located inside an atom. It is the way these particles are arranged that gives each atom its unique characteristics. For example, the arrangement of electrons (also called configuration) decides physical and chemical characteristics. In case of neutral atom, number of electrons is equal to the number of protons. Thus, the number of protons is called the atomic number (Z). The number helps to identify an element in modern periodic table. For example, hydrogen (denoted as H) has atomic number (Z) = 1 and occupies the first position in the table. In the nucleus, Hydrogen has a proton without neutron. This can be considered as special character of the element.